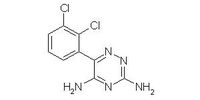
|
已读[47]
发布: 2013/07/30 16:47:51
跌幅榜第一的华海药业(600521)因为FDA新批准两家拉莫三嗪缓释片的ANDA文号而受到市场担忧。 高管等重要股东增减持跟踪 海翔药业(002099)董秘以5.5元均价减持1万...
|
|
|

|
已读[366]
发布: 2013/07/30 16:46:38
与高华证券下调华海药业(600521.SH)目标价不同,中投证券维持5月底给出的6-12个月25元目标价的判断,并强调,公司未来可能有更多像拉莫三嗪这样的品种上市。 2013...
|
|
|

|
已读[398]
发布: 2013/07/30 16:46:15
美国FDA近期批准了2 家抗癫痫药拉莫三嗪缓释片的ANDA,分别是:(1) 6 月19 日批准的印度Dr Reddys的ANDA 文号202383 (规格25/50/100/200/300mg); (2) ...
|
|
|

|
已读[371]
发布: 2013/07/30 16:46:00
美国FDA近期批准了2 家抗癫痫药拉莫三嗪缓释片的ANDA,分别是:(1) 6 月19 日批准的印度Dr Reddys的ANDA 文号202383 (规格25/50/100/200/300mg); (2) ...
|
|
|

|
已读[66]
发布: 2013/07/30 16:45:37
拉莫三嗪新厂家冲击有限,二元竞争格局不变,高盈利能力有望维持 近日Handa和Dr.Reddy’s相继获得拉莫三嗪控释片的ANDA文号。我们认为,Handa由于缺少250和300mg批文...
|
|
|

|
已读[69]
发布: 2013/07/30 16:44:02
而随着Wockhardt因GMP问题或阶段性退出竞争,下半年拉莫三嗪控释片市场或从Par与Wockhardt的二元竞争格局切换为Par与Dr.Reddy’s的二元竞争格局,竞争环境并未显著恶化...
|
|
|

|
已读[99]
发布: 2013/07/30 16:43:57
公司通过多年积累在国外制剂市场已积累了一定市场经验,并且成功打开局面,拉莫三嗪缓释制剂的成功也标志着公司正由低端的普药制剂代加工企业向高端仿制药发展的新战略...
|
|
|

|
已读[32]
发布: 2013/07/30 16:42:41
The Daily Advertiser In addition to Avandia, the settlement also resolves claims relating to the drugs Paxil, Wellbutrin, Advair, Lamictal , Zofran, Imitrex, Lotronex, Flovent and Valtrex for off-label, noncovered uses in violation of state laws, Caldwell's office said. GSK ... and more »
|
|
|

|
已读[37]
发布: 2013/07/30 16:42:24
NOLA.com Louisiana filed a second suit in February, saying the company also illegally promoted Paxil, Wellbutrin, Advair, Lamictal , Zofran, Imitrex, Lotronex, Flovent and Valtrex for off-label uses. A spokeswoman with GSK on Friday said the settlement came at ...
|
|
|

|
已读[36]
发布: 2013/07/30 16:42:07
FOX 8 News WVUE-TV In a news release issued by Caldwell's office, "the settlement also resolves claims relating to the drugs Paxil, Wellbutrin, Advair, Lamictal , Zofran, Imitrex, Lotronex, Flovent and Valtrex for off-label, non-covered uses in violation of state laws." ...
|
|
|
« 上一页 |
查看结果31-40共252
|
下一页 »
|
|