Fine chemicals
产品信息:
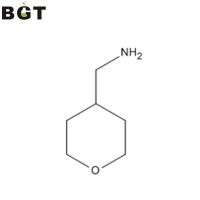
What is 4-Aminomethyltetrahydropyran? 4-Aminomethyltetrahydropyran and its acid salt are useful compounds as a starting material or a synthetic intermediate of medicines, agricultural chemicals, etc.
Application of 4-Aminomethyltetrahydropyran 4-Aminomethyltetrahydropyran is an intermediate for preparing medicines such as Benzimidazole derivatives and Pyrimidine derivatives, which are useful in therapy, particularly in the management of pain.
Ways to prepare 4-Aminomethyltetrahydropyran 1. 4-cyanotetrahydropyran and hydrogen are reacted in the presence of Raney nickel in anhydrous ethanol (International Patent Publication No. 94/05639). However, according to this method, yield of 4-aminomethyltetrahydropyran is low, and a large amount of a by-product (bis(4-tetrahydropyranylmethyl)amine) is produced, so it is not satisfying as an industrial process for preparing 4-aminomethyltetrahydropyran and an acid salt thereof. 2. In an autoclave with an inner volume of 200ml made of stainless equipped with a stirring device, a thermometer, a pressure gauge, and were charged 10.0 g (90.0 mmol) of 4-cyanotetrahydropyran, 50.0 g of 22% by weight ammonia-methanol solution and 2.0 g (17.0 mmol in terms of a nickel atom) of developed Raney nickel (available from Nikki Chemical Co., Ltd.; sponge nickel N154D), and the mixture was reacted under hydrogen atmosphere (0.51 to 0.61MPa) at 45 to 55°C for 17 hours under stirring. After completion of the reaction, insoluble materials were filtered, and the filtrated material was washed with 30 ml of methanol. The filtrate and the washed solution were combined and concentrated under reduced pressure, and then, the concentrate was distilled under reduced pressure (73 to 74°C, 2.67 kPa) to tive 7.94 g (Isolation yield; 76.6%) of 4-aminomethyltetrahydropyran as colorless liquid.
联系我
与我的联系人共享
4-Aminomethyltetrahydropyran MSDS.pdf
Download
供应单标签
4-Aminomethyltetrahydropyran, 130290-79-8
|